Saturday, 6 August 2016
Tuesday, 26 July 2016
Periods and Blocks
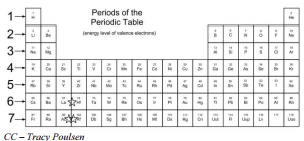
There are seven horizontal rows of the periodic table, called periods. The length of each period is determined by the number of electrons that are capable of occupying the sublevels that fill during that period, as seen in the table below.
Recall that the four different sublevels each consist of a different number of orbitals. The
The period to which a given element belongs can easily be determined from its electron configuration. For example, consider the element nickel
Based on electron configurations, the periodic table can be divided into blocks denoting which sublevel is in the process of being filled. The
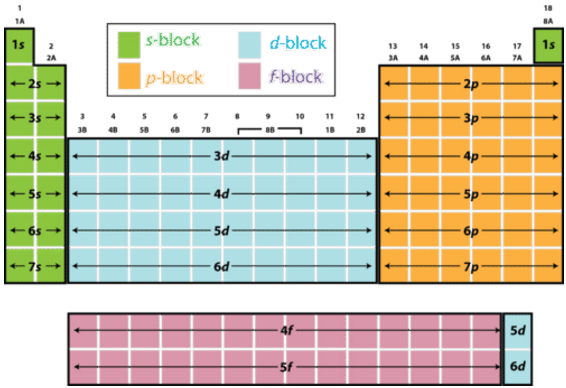
Figure 6.8.1: The periodic table labeled with sublevel blocks.
The figure also illustrates how the
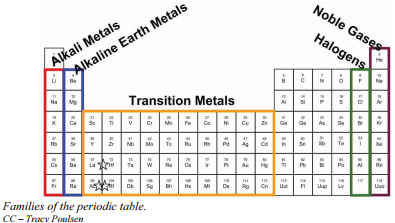
Families of the Periodic Table
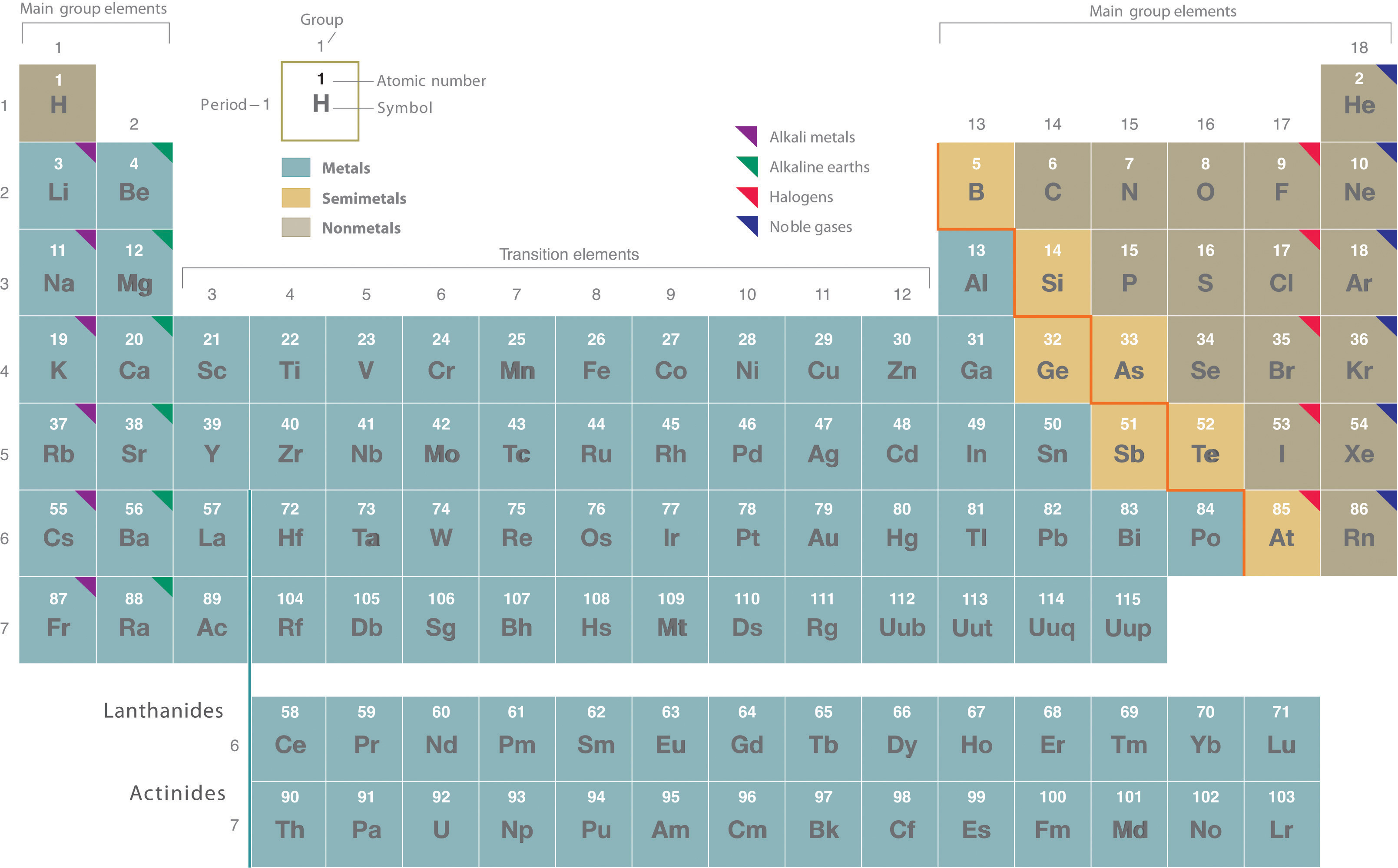
Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. A group is a vertical column of the periodic table. All of the 1A elements have one valence electron. This is what causes these elements to react in the same ways as the other members of the family. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. Group 1A is also known as the alkali metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut.
Group 2A is also called the alkaline earth metals. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. The same pattern is true of other groups on the periodic table. Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table.
It is important to recognize a couple of other important groups on the periodic table by their group name. Group 7A (or 17) elements are also called halogens. This group contains very reactive nonmetal elements.
The noble gases are in group 8A. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. We will learn the reason for this later, when we discuss how compounds form. The elements in this group are also gases at room temperature.
An alternate numbering system numbers all of the
Periods of the Periodic Table
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group numbera | [hide]1 | 2 | 3d | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mendeleev (I–VIII) | I | II | III | IV | V | VI | VII | VIII | I | II | III | IV | V | VI | VII | b | ||||
| CAS (US, pattern A-B-A) | IA | IIA | IIIB | IVB | VB | VIB | VIIB | VIIIB | IB | IIB | IIIA | IVA | VA | VIA | VIIA | VIIIA | ||||
| old IUPAC (Europe, pattern A-B) | IA | IIA | IIIA | IVA | VA | VIA | VIIA | VIII | IB | IIB | IIIB | IVB | VB | VIB | VIIB | 0 | ||||
| Trivial name | Alkali metals | Alkaline earth metals | Coinage metalse | Volatile metalse | Icosagense | Crystallogense | Pnictogens | Chalcogens | Halogens | Noble gases | ||||||||||
| Name by element | Lithium group | Beryllium group | Scandium group | Titanium group | Vanadium group | Chromium group | Manganese group | Iron group | Cobalt group | Nickel group | Copper group | Zinc group | Boron group | Carbon group | Nitrogen group | Oxygen group | Fluorine group | Helium orNeon group | ||
| Period 1 | Hc | He | ||||||||||||||||||
| Period 2 | Li | Be | B | C | N | O | F | Ne | ||||||||||||
| Period 3 | Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||
| Period 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||
| Period 5 | Rb | Sr | d | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |
| Period 6 | Cs | Ba | La–Yb | Lud | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |
| Period 7 | Fr | Ra | Ac–No | Lrd | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | |
| a Current, modern IUPAC group number. | ||||||||||||||||||||
| b The noble gases had not yet been discovered at the time of Mendeleev's original table. Later (1902), Mendeleev accepted the evidence for the existence of the noble gases, and placed them in a separate "group 0". | ||||||||||||||||||||
| c Hydrogen (H), while placed in column 1, is not considered to be in the group alkali metals. | ||||||||||||||||||||
| d Group 3: Depending on the source, this group includes lutetium (Lu) and lawrencium (Lr), or lanthanum (La) and actinium (Ac), or the whole set of 15 lanthanides and 15 actinides, as the entries below scandium and yttrium. | ||||||||||||||||||||
| e This group name is not recommended by IUPAC. | ||||||||||||||||||||













Subscribe to:
Comments (Atom)